The Haber Process
Chemical Equilibria|Le Chatelier's Principle|Factors Affecting Chemical equilibria| The Haber Process|The Contact Process|Equilibrium Constants
The Haber Process
The Haber process is the name given to the process during which ammonia is manufactured.
During this reaction, Nitrogen obtained from the fractional distillation of liquid air is combined with Hydrogen obtained from cracking of hydrocarbons or from methane gas.
The manufacture of ammonia is a reversible reaction.
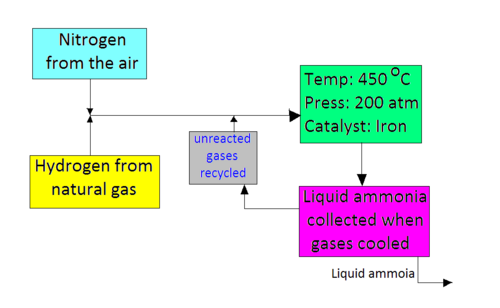
The following shows a diagramatic representation of the process:
As seen from the diagram, the required conditions for the reaction are:
- Temperature: 450 oC
- Pressure: 200 atm
- Catalyst: Iron Filings
N2(g) + 3H2(g) <=> 2NH3(g) ∆H = -92 kJ/mol
Temperature
A temperature of 450 oC is used during this reaction. As observed, the enthalpy change for the reaction is exothermic. As a result, a low temperature would have favoured the forward reaction, thus increasing the yield of ammonia prodcued. However, a too low temperature cannot be used because the rate of the reaction would be slowed down considerably. Thus a compromise temperature of 450 oC is used which is high enough for rate to be quite fast and low enough to get a relatively high yield of ammonia.
Pressure
A pressure of 200 atm is used for this reaction. According to Le Chatelier's Principle, if we increase the pressure, the side with the lower number of moles would be favoured. This implies that if pressure is increased, the forward reaction would be favoured, producing more ammonia.
However, this cannot be done because it involves a very high health risk. At this very high pressure, it becomes dangerous for the people working in the Haber plant. Moreover, the cost of securing the work area becomes enormous and much money has to be spent in this area. To decrease the risk factor as well as the cost, a compromise pressure of 200 atm is used.
Catalyst
Iron fillings is used as catalyst in this reaction to increase the rate of the reaction because a higher temperature cannot be used.