Video
Alkali Metals
- The alkali metals provide one of the best examples of group trends in properties in the periodic table, with well characterized homologous behavior down the group.
- The alkali metals are all highly reactive and are rarely found in elemental form in nature.
- In the laboratory they are stored under mineral oil.
- They also tarnish easily and have low melting points and densities.
- Potassium and rubidium possess a weak radioactive characteristic (harmless) due to the presence of long duration radioactive isotopes.
- They are silver-colored (caesium has a golden tinge)
- They are soft, low-density metals.
- They react readily with halogens to form ionic salts, and with water to form strongly alkaline (basic) hydroxides.
- These elements all have one electron in their outermost shell, so the energetically preferred state of achieving a filled electron shell is to lose one electron to form a singly charged positive ion, i.e. cation.
Alkaline Earth Metals
- The alkaline earth metals provide a good example of group trends in properties in the periodic table, with well characterized homologous behavior down the group.
- The alkaline earth metals are silvered colored, soft metals.
- They react readily with halogens to form ionic salts, and with water, though not as rapidly as the alkali metals, to form strongly alkaline (basic) hydroxides. For example, where sodium and potassium react with water at room temperature, magnesium reacts only with steam and calcium with hot water:
Mg + 2 H2O → Mg(OH)2 + H2
- Beryllium is an exception: It does not react with water or steam, and its halides are covalent.
- All the alkaline earth metals have two electrons in their valence shell, so the energetically preferred state of achieving a filled electron shell is to lose two electrons to form doubly charged positive ions.
- The alkaline earth metals are named after their oxides. These oxides are basic (alkaline) when combined with water.
Noble gases
- These are elements found in Group 8.
- They have full valence shells. This means they do not need to react with other elements to attain a full shell.
- They are less reactive than other groups.
- Neon followed by helium are the most inert elements among noble gases.
- Reactivity, in this group, increases with the periods: it is possible to make heavy noble gases react since they have much larger electron shells. However, their reactivity remains very low in absolute terms.
Halogens
- These are elements found in Group 7.
- These elements are missing just one electron each to fill their shells.
- In chemical reactions they tend to acquire electrons (the tendency to acquire electrons is called electronegativity). This property is most evident for fluorine (the most electronegative element of the whole table), and it diminishes with increasing period.
- All halogens form acids with hydrogen (all in the form HX), such as:
- hydrofluoric acid
- hydrochloric acid
- hydrobromic acid
- hydroiodic acid.
- Their acidity increases with higher period, for example, with regard to iodine and fluorine, since a large I− ion is more stable in solution than a small F−, there is less volume in which to disperse the charge.
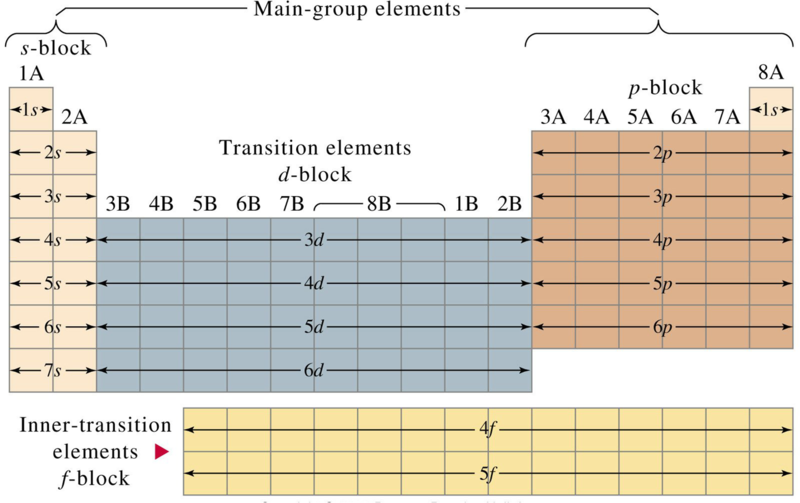
Transition metals
- For the transition metals (found between Group II and Group III), horizontal trends across periods are often important as well as vertical trends down groups.
- the differences between groups adjacent are usually not dramatic.
- Transition metal reactions often involve coordinated species.
Lanthanides and actinides
- All lanthanides closely resemble lanthanum.
- They are electropositive trivalent metals.
- They are shiny and silvery-white, and tarnish easily when exposed to air. Many make steel.
- They react violently with most nonmetals.
- They are relatively soft but their hardness increases with their atomic number.
- Lanthanides burn in air.
- They have high melting and boiling points.
- Most lanthanides are widely used in lasers. These elements deflect UV and Infrared electromagnetic radiation and are commonly used in the production of sunglass lenses.
- Due to their specific electronic configurations, lanthanide atoms tend to lose three electrons to attain their most stable oxidation state as trivalent ions.
|